🫁 Supporting Respiratory Drug Discovery at Pharmidex
January 15, 2026
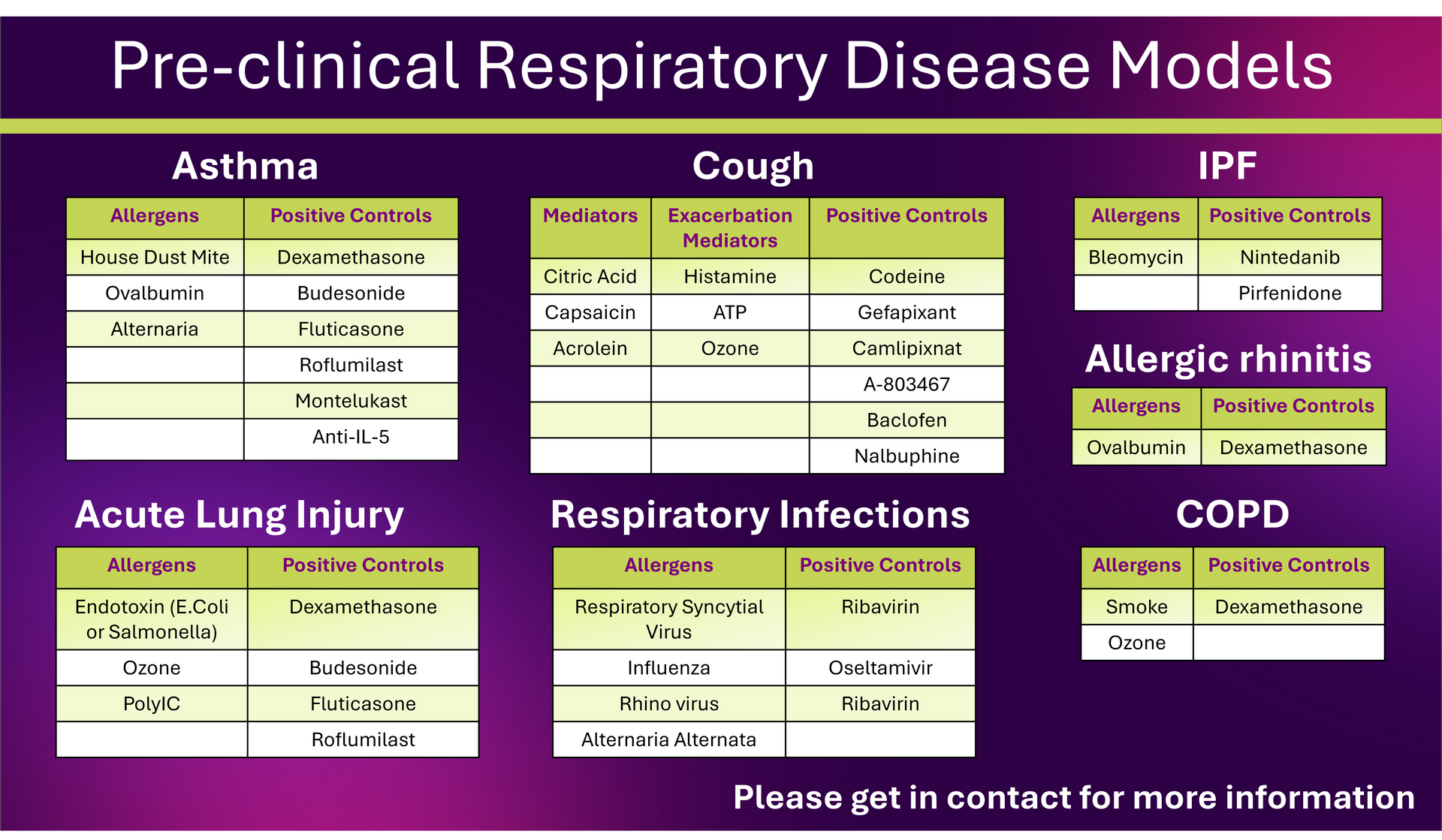
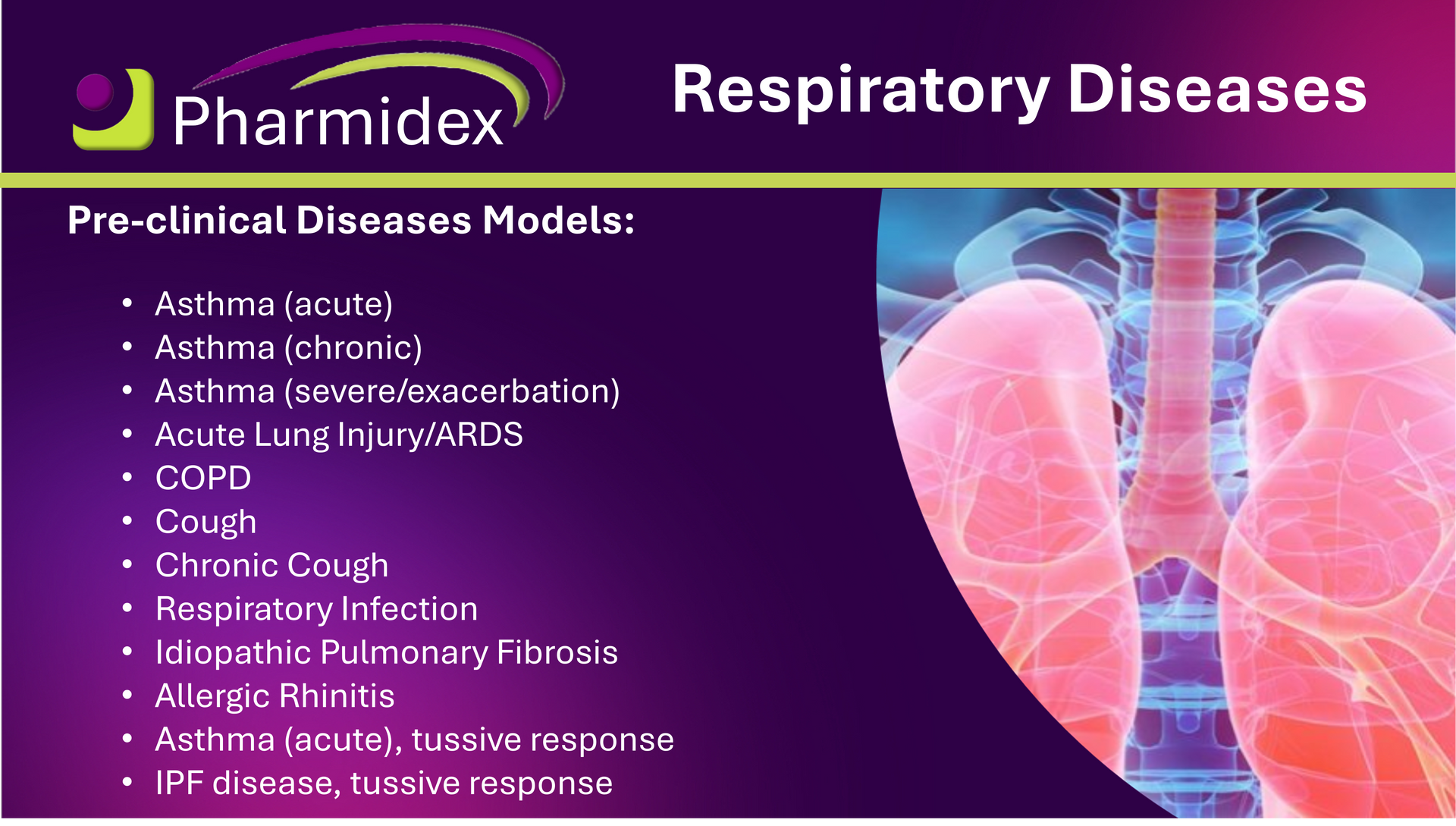
Pharmidex offers a broad range of pre-clinical respiratory disease models to support drug discovery and development across multiple pulmonary indications.
Our capabilities include validated models for asthma (acute, chronic and severe/exacerbation), cough, idiopathic pulmonary fibrosis (IPF), acute lung injury/ARDS, respiratory infections, COPD and allergic rhinitis.
These models incorporate clinically relevant triggers and well-established positive controls, enabling robust and translational assessment of novel therapeutics.
Our integrated respiratory platforms are designed to help accelerate informed decision-making from early research through to advanced pre-clinical development.
If you are developing new therapies for respiratory diseases, we would be pleased to discuss how Pharmidex can support your programme.
🌐
www.pharmidex.com

What started in 2002 with a small team and a handful of clients has grown into a global preclinical CRO trusted by more than 400 clients worldwide, including many leading pharmaceutical and biotechnology companies. Our commitment to delivering bespoke and standard study designs in a quick, expert, and cost-effective manner has allowed us to support more than 1,000 studies per year for multiple consecutive years, something we are incredibly proud of. Over the years, we have been fortunate to contribute data supporting numerous marketed drugs, numerous successful client acquisitions, and programmes successfully supporting patients. Most importantly, our success is a testament to the wider scientific community and the innovative ideas and projects our clients trust us to support every day. Thank you to every client, collaborator, colleague, and friend who has been part of our journey so far. We look forward to supporting the next generation of science and innovation for another 24 years.

We are delighted to announce the promotion of Dr. Martin Barrett to Chief Business Officer at Pharmidex . Martin brings more than 23 years of experience across the pharmaceutical and drug discovery sectors, combining deep scientific expertise with strategic commercial leadership. Throughout his career, he has played a key role in advancing translational science, building strategic partnerships and supporting innovative drug development programmes. In his new role, Martin will help drive Pharmidex’s continued growth, strengthen collaborations, and expand our capabilities across preclinical pharmacology, toxicology, translational science, and clinical development support. Congratulations to Martin on this well-deserved promotion. We look forward to the continued impact of his leadership across Pharmidex and the wider drug discovery community.

Pharmidex is pleased to announce that Janette Dalay Robertson , Business Development Manager, will be attending the Milner Seminar May 2026 hosted by the Milner Therapeutics Institute at the Jeffrey Cheah Biomedical Centre , Cambridge, on 19 May 2026 from 4:00–5:00pm. The seminar will bring together experts from academia and industry to discuss innovation and collaboration in drug discovery and translational science. Pharmidex will also be exhibiting at the event. Janette will be near the café in the Jeffrey Cheah Biomedical Centre from 11am tomorrow, so if you would like to discuss drug discovery, preclinical development, bioanalysis, or potential collaborations, please feel free to stop by for a chat before the seminar begins. Looking forward to connecting with researchers, biotech innovators, and industry partners throughout the day!